Maternal blood collection
10 mL Streck/G-tube — label and ship promptly.
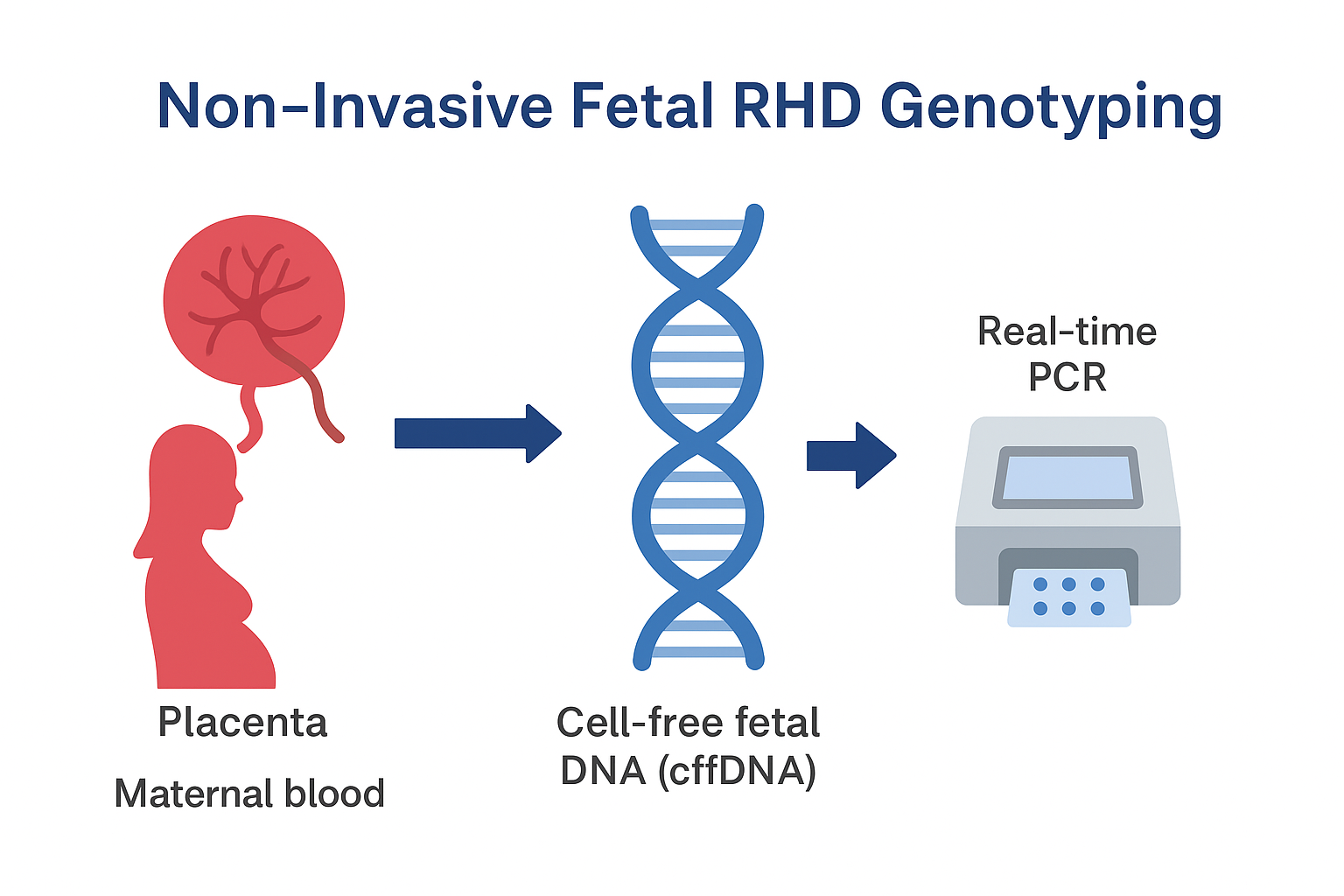
Non-invasive foetal RHD genotyping from maternal plasma — clinical utility, workflow and reporting guidance.
Reliable from ≥10 weeks gestation; accuracy >99% with validated assays.Placenta → cffDNA in maternal plasma → extraction → PCR targets → interpretation
| Result | Meaning |
|---|---|
| Foetal RHD detected | Fetus predicted RhD-positive. Anti-D prophylaxis recommended. |
| Foetal RHD not detected | Foetus predicted RhD-negative. Anti-D may not be required. |
| Inconclusive | Low foetal fraction or interference — repeat sample suggested. |